The molecular basis for the large (and perhaps exclusive) preference for Arg over Lys as a gating charge carrier in VSD has not been established. The mobile charges on S4 that pass through the hydrophobic plug (gating charges) are almost exclusively carried on arginine side chains indeed it is possible that lysine is never a carrier of mobile charges across the hydrophobic plug in membrane voltage sensors. In voltage-mediated transitions the positive side chains pass through a hydrophobic “plug” 7 or “gasket” 8 which separates the internal and external regions of the VS and provides a barrier to passage of ions, focusing the membrane potential across a narrow region of the membrane. The S4 helix contains a repeating triplet motif consisting of positively charged amino acids (Lys or Arg) separated by two hydrophobic amino acids it is the interaction of S4 positive charges with the membrane potential that drives the conformational changes underlying membrane voltage sensitivity. This is particularly marked for the voltage-sensor domains (VSD) of voltage sensitive ion channels and enzymes that contain a mobile transmembrane helical segment (S4) which moves up and down through the membrane plane as the VSD responds to changes in membrane potential 5, 6. The physicochemical properties of these side chains underlie the roles of Arg and Lys in diverse biological structures and functions. This contrasts with Lys where the charge is largely focused on the terminal aliphatic amino group and the side chain is readily deprotonated in proteins 1. The very high pKa of Arg results from delocalization of the positive charge within the π-bonded system of the side chain guanidinium ion so stable is the guanidinium that the Arg side chain remains protonated when partially buried within protein structures or membranes 3, 4. These amino acids have profound roles in protein structure and function that involve electrostatic interactions and protein solvation. Lysine and arginine are the two positively charged amino acids in proteins that have high aqueous pKa’s (~10.5 for Lys 1 and ~13.8 for Arg 2) indicating a strong propensity to carry charge at physiological pH. The hydration properties of arginine and lysine are strongly reflected in their respective interactions with non-polar and polar side chains as observed in protein structures and in molecular dynamics simulations and likely underlie the preference for arginine as a mobile charge carrier in VSD. In contrast, non-polar side chains interact largely with the aliphatic part of the lysine side chain. The arginine guanidinium interacts with non-polar aromatic and aliphatic side chains above and below the guanidinium plane while hydrogen bonding with polar side chains is restricted to in-plane positions. To test for side chain interactions implicit in this model we inspected interactions of the side chains of arginine and lysine with each of the 19 non-glycine amino acids in proteins in the protein data bank. This arginine preference may result from the unique hydration properties of the side chain guanidinium group which facilitates its movement through a hydrophobic plug that seals the center of the VSD, as suggested by molecular dynamics simulations.
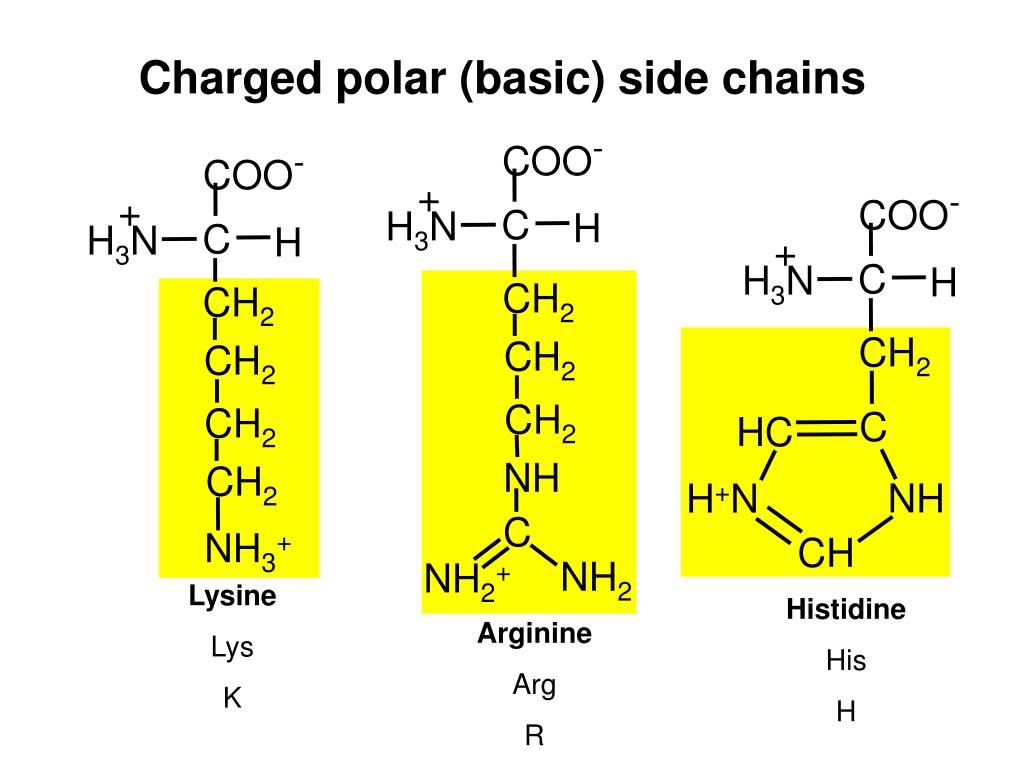
Gating charges in voltage-sensing domains (VSD) of voltage-sensitive ion channels and enzymes are carried on arginine side chains rather than lysine.
